Bacteria: Gram Staining and Antibiotics
In 1884 Hans Christian Gram, a Danish bacteriologist, attempted to find a universal stain that would work with all bacteria. In the process, he discovered that bacteria could be divided into two different groups -- one that retained a stain, called "gram-positive," and one that didn't, called "gram-negative." His unique method for identifying these two groups became the first step in any bacterial identification process. Even the simple determination that a bacteria specimen is gram-positive or gram- negative can direct a doctor in diagnosis, as different bacteria cause different diseases. For example, the bacteria that causes scarlet fever is gram-positive, while that which causes typhoid or cholera is gram-negative.
First Grow bacteria on nutrient agar plate.
First Grow bacteria on nutrient agar plate.
Purpose: to Gram Stain a bacterial sample and observe and draw them
Part One - Gram Stain. You will not actually do it but need to know how it is done for test purposes
Materials
· Live bacteria cultures - Bacillus Cereus and Rodospirillum Rubrum. (You could also grow your owncultures with agarand petri dishes)
· Inoculating needle
· Gram stain kit(contains crystal violet stain, Gram iodine stain, ethyl alcohol solvent, Safranin O counterstain, plain microscope slides, medicine dropper, coverslips)
· Wash bottle
· Compound Microscope
Procedure
Some of the steps of the gram stain process are hard to carry out perfectly. To practice, it is a good idea to make a "control" slide. Try collecting some bacteria from between your teeth (using a toothpick) and placing it on a slide with a drop of water. If the Gram staining procedure is done correctly, your slide should have a mixture of gram-negative and gram-positive cells as well as some neutrophils (white blood cells) with pink nuclei. After you have tried that, stain each of your live bacteria cultures using the following procedure:
1. Sterilize your inoculating needle by placing it in a candle flame. Let it cool for 3-5 seconds.
2. Make a specimen smear by placing a small amount of bacteria from one of the cultures on a clean glass slide with the inoculating needle. Take another slide and use its edge to scrape or "smear" the specimen into a very thin film of material.
3. Let the specimen on the slide air dry, and then heat fix it by passing the slide through a candle flame 3-4 times. (The slide shouldn't get too hot to touch, and it should never stop as it passes through the flame.)
4. Cover the specimen with 1-2 drops of the crystal violet stain for 60 seconds and then gently wash it off with very slow running water from the tap or a few gentle squirts from a wash bottle. (If the water is running too fast and hits the slide with too much force, the specimen will be washed off.)
5. Cover the specimen with a few drops of Gram's iodine for 60 seconds; then gently wash the specimen again as in step 4.
6. Use ethyl alcohol as the solvent. This is the most sensitive step, because if the ethyl alcohol is left on the specimen too long, it will decolorize the gram-positive cells as well as the gram-negative. Tilt the slide slightly and apply the alcohol drop by drop onto the slide above the specimen, so that the alcohol runs down over the entire specimen. Stop applying the alcohol when the fluid flowing off the edge of the slide is no longer colored. The thinnest parts of the smear should be colorless. This will take about 5 seconds. Wash the slide gently again. Note that gram-positive cells will retain some of the violet coloring, but the majority of the stain will be rinsed away by the solvent.
7. Cover the specimen with a few drops of safranin stain as the counter stain for 60 seconds and then gently wash once more.
8. Blot the slide with absorbent paper (a paper towel will work if you have nothing else), but do not rub the specimen smear. Put a coverslip over the smear.
9. Now you are ready to examine your slide under a microscope at each magnification level. As you do so, look for cells that are purple in color. These are gram positive cells that retained the crystal violet stain. Cells that are pink or red in color are gram negative cells. In these cells, the crystal violet was washed away by the ethyl alcohol and replaced with the safranin.
For the makeup lab find the poster in the room of bacteria that have been Gram Stained and draw and label all of them. Make a table with name, shape, Gram Stain colour, arrangement of the bacteria & special feature*
Here are the photos from the poster
Part One - Gram Stain. You will not actually do it but need to know how it is done for test purposes
Materials
· Live bacteria cultures - Bacillus Cereus and Rodospirillum Rubrum. (You could also grow your owncultures with agarand petri dishes)
· Inoculating needle
· Gram stain kit(contains crystal violet stain, Gram iodine stain, ethyl alcohol solvent, Safranin O counterstain, plain microscope slides, medicine dropper, coverslips)
· Wash bottle
· Compound Microscope
Procedure
Some of the steps of the gram stain process are hard to carry out perfectly. To practice, it is a good idea to make a "control" slide. Try collecting some bacteria from between your teeth (using a toothpick) and placing it on a slide with a drop of water. If the Gram staining procedure is done correctly, your slide should have a mixture of gram-negative and gram-positive cells as well as some neutrophils (white blood cells) with pink nuclei. After you have tried that, stain each of your live bacteria cultures using the following procedure:
1. Sterilize your inoculating needle by placing it in a candle flame. Let it cool for 3-5 seconds.
2. Make a specimen smear by placing a small amount of bacteria from one of the cultures on a clean glass slide with the inoculating needle. Take another slide and use its edge to scrape or "smear" the specimen into a very thin film of material.
3. Let the specimen on the slide air dry, and then heat fix it by passing the slide through a candle flame 3-4 times. (The slide shouldn't get too hot to touch, and it should never stop as it passes through the flame.)
4. Cover the specimen with 1-2 drops of the crystal violet stain for 60 seconds and then gently wash it off with very slow running water from the tap or a few gentle squirts from a wash bottle. (If the water is running too fast and hits the slide with too much force, the specimen will be washed off.)
5. Cover the specimen with a few drops of Gram's iodine for 60 seconds; then gently wash the specimen again as in step 4.
6. Use ethyl alcohol as the solvent. This is the most sensitive step, because if the ethyl alcohol is left on the specimen too long, it will decolorize the gram-positive cells as well as the gram-negative. Tilt the slide slightly and apply the alcohol drop by drop onto the slide above the specimen, so that the alcohol runs down over the entire specimen. Stop applying the alcohol when the fluid flowing off the edge of the slide is no longer colored. The thinnest parts of the smear should be colorless. This will take about 5 seconds. Wash the slide gently again. Note that gram-positive cells will retain some of the violet coloring, but the majority of the stain will be rinsed away by the solvent.
7. Cover the specimen with a few drops of safranin stain as the counter stain for 60 seconds and then gently wash once more.
8. Blot the slide with absorbent paper (a paper towel will work if you have nothing else), but do not rub the specimen smear. Put a coverslip over the smear.
9. Now you are ready to examine your slide under a microscope at each magnification level. As you do so, look for cells that are purple in color. These are gram positive cells that retained the crystal violet stain. Cells that are pink or red in color are gram negative cells. In these cells, the crystal violet was washed away by the ethyl alcohol and replaced with the safranin.
For the makeup lab find the poster in the room of bacteria that have been Gram Stained and draw and label all of them. Make a table with name, shape, Gram Stain colour, arrangement of the bacteria & special feature*
Here are the photos from the poster
1 Neisseria gonorrhea
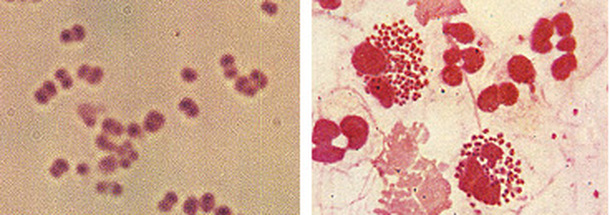
2 Streptococcus pneumoniae
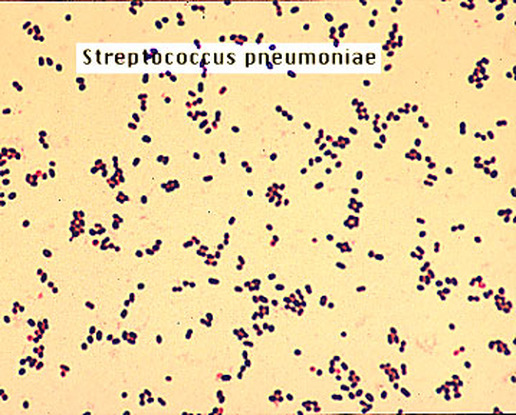
3 Streptococcus pyogenes
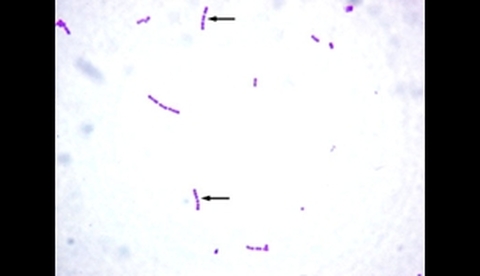
4 Staphylococcus epidermidis
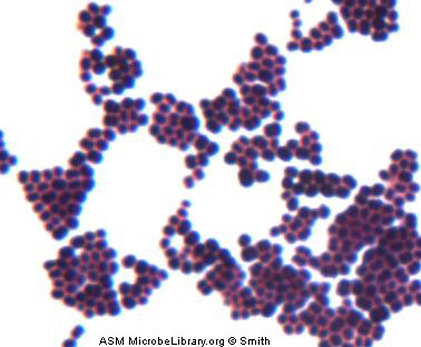
5 Escherichia coli
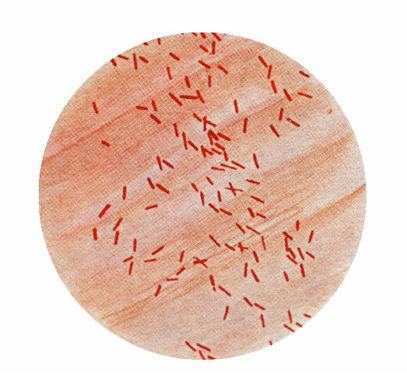
6 Proteus vulgaris(*flagella)
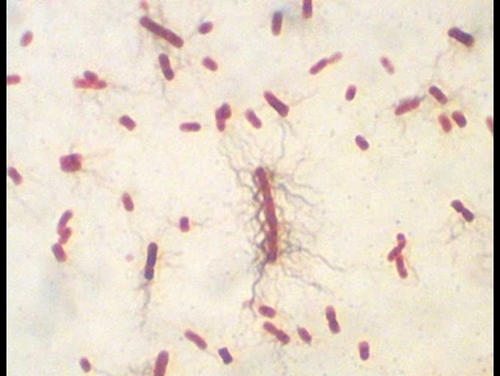
7 Clostridium botulinum (*spores- ones with oval clear spots)
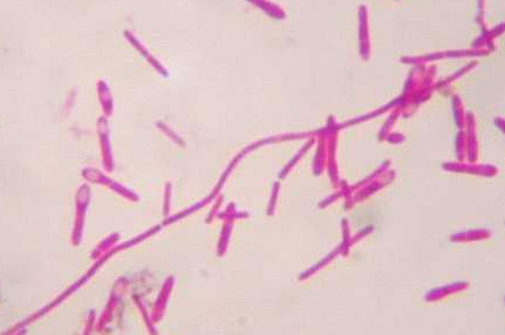
8 Klebsiella pneumoniae(*capsule- clear area around bacteria)
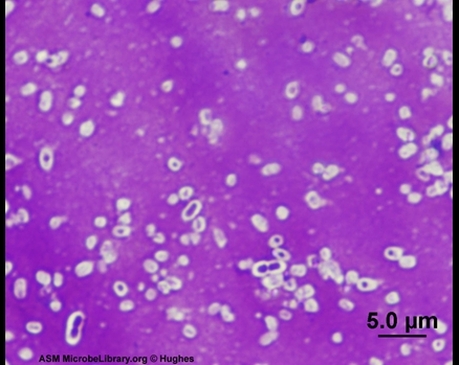
9 Bacillus subtilis
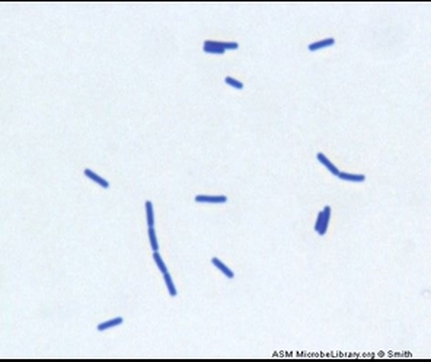
10 Actinomyces
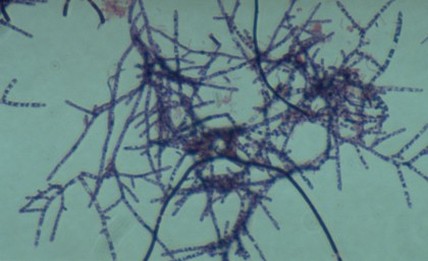
11 Vibrio cholerae
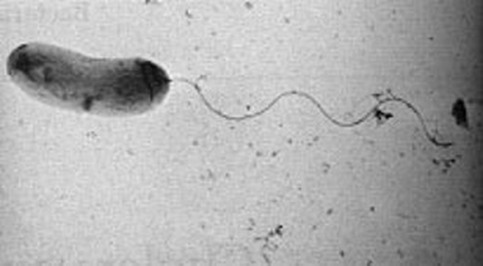
12 Rhodospirillum rubrum
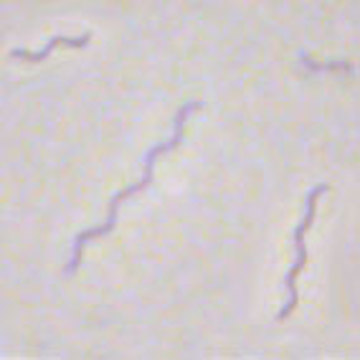
13 Spirillum volutans(*flagella)
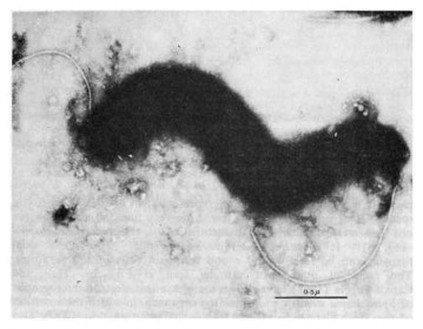
14 Treponema pallidum
Part Two - Antibiotic Testing
Purpose :to measure the zones of inhibition around antibiotic disks and compare them to one another for makeup lab you will not actually do this procedure but you need to know how it is done
One way to test bacteria susceptibility to antibiotics is to use the Kirby-Bauer or "disc diffusion" method. This method involves measuring the inhibition of bacteria growth around an antibiotic disc placed in a culture.
Materials
Procedure
1. Prepare the agar according to the directions on the label, then pour 10-15 ml into each petri dish (enough to cover the bottom of the dish). Let the dish stand for about an hour until the agar is firm.
2. Sterilize your inoculating needle and then inoculate one dish with the gram-positive bacteria. Lightly zig-zag the needle over the surface of the agar, turn the dish, and do it again. Do this several times to get maximum distribution.
3. Place one disc of each antibiotic type at different places on the agar (use sterile tweezers). Press the disc down slightly to secure it in the agar. Cover the dish when you're done.
4. Repeat steps 2-3 with the gram-negative bacteria.
5. Examine each dish after 24 hours. If the bacteria culture grows right up to the edge of the antibiotic disc, it is not susceptible to that antibiotic. If there is a circular area around the disc where the bacteria growth is inhibited, measure and record the diameter of the circle. Make note of the effect of each antibiotic disc in each petri dish. You may also wish to take pictures.
6. Repeat step 5 after 48 hours.
Plate 1
Purpose :to measure the zones of inhibition around antibiotic disks and compare them to one another for makeup lab you will not actually do this procedure but you need to know how it is done
One way to test bacteria susceptibility to antibiotics is to use the Kirby-Bauer or "disc diffusion" method. This method involves measuring the inhibition of bacteria growth around an antibiotic disc placed in a culture.
Materials
Procedure
1. Prepare the agar according to the directions on the label, then pour 10-15 ml into each petri dish (enough to cover the bottom of the dish). Let the dish stand for about an hour until the agar is firm.
2. Sterilize your inoculating needle and then inoculate one dish with the gram-positive bacteria. Lightly zig-zag the needle over the surface of the agar, turn the dish, and do it again. Do this several times to get maximum distribution.
3. Place one disc of each antibiotic type at different places on the agar (use sterile tweezers). Press the disc down slightly to secure it in the agar. Cover the dish when you're done.
4. Repeat steps 2-3 with the gram-negative bacteria.
5. Examine each dish after 24 hours. If the bacteria culture grows right up to the edge of the antibiotic disc, it is not susceptible to that antibiotic. If there is a circular area around the disc where the bacteria growth is inhibited, measure and record the diameter of the circle. Make note of the effect of each antibiotic disc in each petri dish. You may also wish to take pictures.
6. Repeat step 5 after 48 hours.
Plate 1
When you are finished observing your bacteria cultures, put in a plastic autoclave bag, autoclave, cool and throw them away.
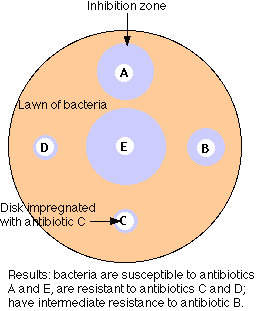
The figure illustrates the simplest method of the several available for measuring antibiotic resistance. A suspension of the bacteria to be tested (e.g. cultured from the infected patient) is spread over the surface of a petri dish containing a solid culture medium.
The figure illustrates the simplest method of the several available for measuring antibiotic resistance. A suspension of the bacteria to be tested (e.g. cultured from the infected patient) is spread over the surface of a petri dish containing a solid culture medium.
- Disks of several different antibiotics are pressed on the surface of the agar. The concentration of antibiotic in each type of disk is standardized.
- Incubate overnight.
- The bacteria will grown into a "lawn" except where an antibiotic to which they are sensitive has diffused out from its disk.
- Measure the diameter of any zones of inhibition that are formed.
Plate 2
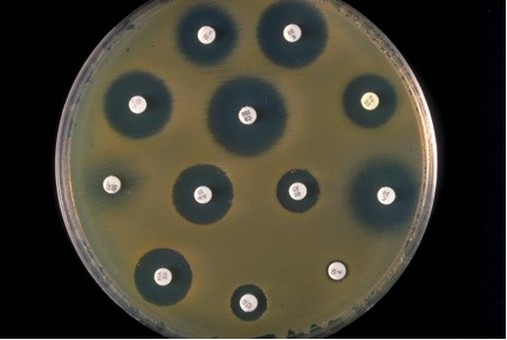
Plate 3
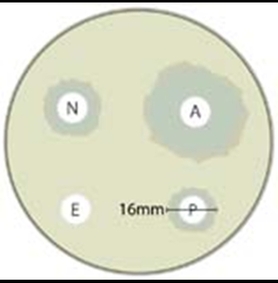
Plate 4
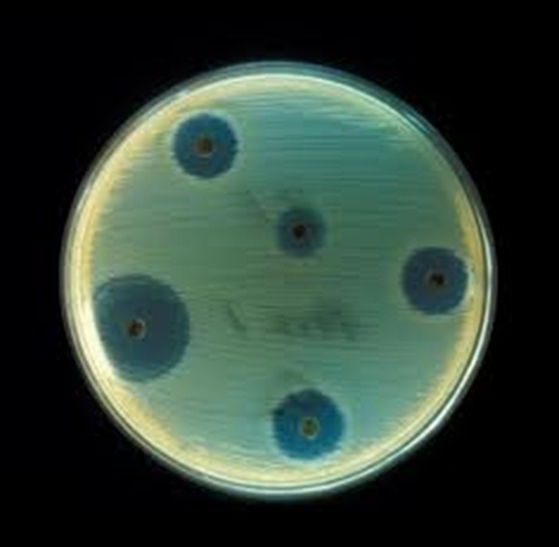
Plate 5
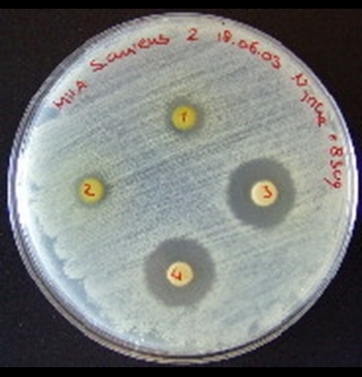
Makeup lab
Results: Make a table showing the zones of inhibition for each of the disks (try to read the symbols on the disk) on each plate
If you can't read the letters then sketch each plate and number off the disks.
Assume each plate is the same size
. Assume the pictures have been scaled sucessfully so that the disks are the same size each time.
Conclusion: which is the best to worst antibiotic for this bacteria? Rank all of them.
Results: Make a table showing the zones of inhibition for each of the disks (try to read the symbols on the disk) on each plate
If you can't read the letters then sketch each plate and number off the disks.
Assume each plate is the same size
. Assume the pictures have been scaled sucessfully so that the disks are the same size each time.
Conclusion: which is the best to worst antibiotic for this bacteria? Rank all of them.