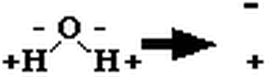Compounds notes
B. CELL COMPOUNDS:
B1&2. WATER
B1&2. WATER
a) not organic (No Carbon)
b) polar molecule, (one side slightly - other side +) due to uneven electron sharing
slight attraction between + and - sides of adjacent molecules
resulting attraction is "sticky" and is called a Hydrogen Bond
c) boils at 100 degrees, freezes at 0 degrees.
d) surface tension - surface is sticky
e) can absorb lot of heat f) liquid is more dense than the solid (ice floats)
g) dissolves a larger number of chemical, particularly polar chemicals.
B3. ACIDS & BASES
pH, Acids, Bases and Buffers:
Acids: - any compound when dissolved in water, releases H+ (Hydrogen) ions.
ex: HCl H+ + Cl-
- This reaction is almost complete therefore
the HCl is a strong acid.
Bases: - compounds that dissolve in water and release OH- (Hydroxyl Ions)
ex: NaOH Na+ + OH-
- This reaction is almost complete therefore the NaOH is a strong base.
pH: - a scale to measure the strength of acids and bases
- ranges from 0 to 14
- 7 is neutral (distilled water)
- pH is the negative log of the H+ (Hydrogen ion) concentration.
- a pH of 2 is ten times stronger than a pH of 3.
Buffers: A chemical (or combination of) mixture of chemicals that "soaks up" H+ or OH- ions.
They maintain a constant pH
- buffers are not permanent
- Can only hold so much H+ or OH-
- Eventually overwhelmed (can run out)
*Important: inside cells
blood
lakes (acid rain)
b) polar molecule, (one side slightly - other side +) due to uneven electron sharing
slight attraction between + and - sides of adjacent molecules
resulting attraction is "sticky" and is called a Hydrogen Bond
c) boils at 100 degrees, freezes at 0 degrees.
d) surface tension - surface is sticky
e) can absorb lot of heat f) liquid is more dense than the solid (ice floats)
g) dissolves a larger number of chemical, particularly polar chemicals.
B3. ACIDS & BASES
pH, Acids, Bases and Buffers:
Acids: - any compound when dissolved in water, releases H+ (Hydrogen) ions.
ex: HCl H+ + Cl-
- This reaction is almost complete therefore
the HCl is a strong acid.
Bases: - compounds that dissolve in water and release OH- (Hydroxyl Ions)
ex: NaOH Na+ + OH-
- This reaction is almost complete therefore the NaOH is a strong base.
pH: - a scale to measure the strength of acids and bases
- ranges from 0 to 14
- 7 is neutral (distilled water)
- pH is the negative log of the H+ (Hydrogen ion) concentration.
- a pH of 2 is ten times stronger than a pH of 3.
Buffers: A chemical (or combination of) mixture of chemicals that "soaks up" H+ or OH- ions.
They maintain a constant pH
- buffers are not permanent
- Can only hold so much H+ or OH-
- Eventually overwhelmed (can run out)
*Important: inside cells
blood
lakes (acid rain)